Calculate the mass of one molecule of carbon dioxide atomic mass of carbon and oxygen are 12 and 16 - Brainly.in

What is the mass of carbon dioxide which contains the same number of molecules as are contained in 40 g of oxygen?

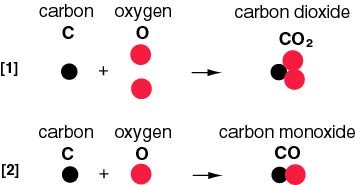
What is the Molar Mass of Carbon Monoxide? and what is the molecular weight of carbon Dioxide? - YouTube

SOLVED: What is the molar mass of carbon monoxide (CO)? Atomic weight of carbon is 12 and oxygen is 16. Answer: g/mol How many moles of CO gas if its mass is

Convert 22 g of carbon dioxide `(CO_(2))` into moles. (Atomic masses : `C = 12 u, O = 16 u`) - YouTube
![Calculate the mass of 0.1 mole of carbon dioxide. [Atomic mass : S = 32, C = 12 and O = 16 and Avogadro.s Number = 6 xx 10^(23)] Calculate the mass of 0.1 mole of carbon dioxide. [Atomic mass : S = 32, C = 12 and O = 16 and Avogadro.s Number = 6 xx 10^(23)]](https://d10lpgp6xz60nq.cloudfront.net/web-thumb/644026465_web.png)



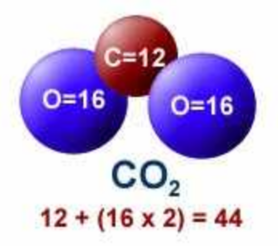


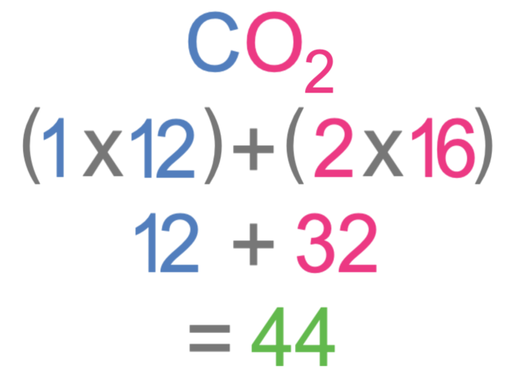


![Carbon Monoxide [CO] Molecular Weight Calculation - Laboratory Notes Carbon Monoxide [CO] Molecular Weight Calculation - Laboratory Notes](https://www.laboratorynotes.com/wp-content/uploads/2021/11/carbon-dioxide-molecular-weight-calculation.jpg)