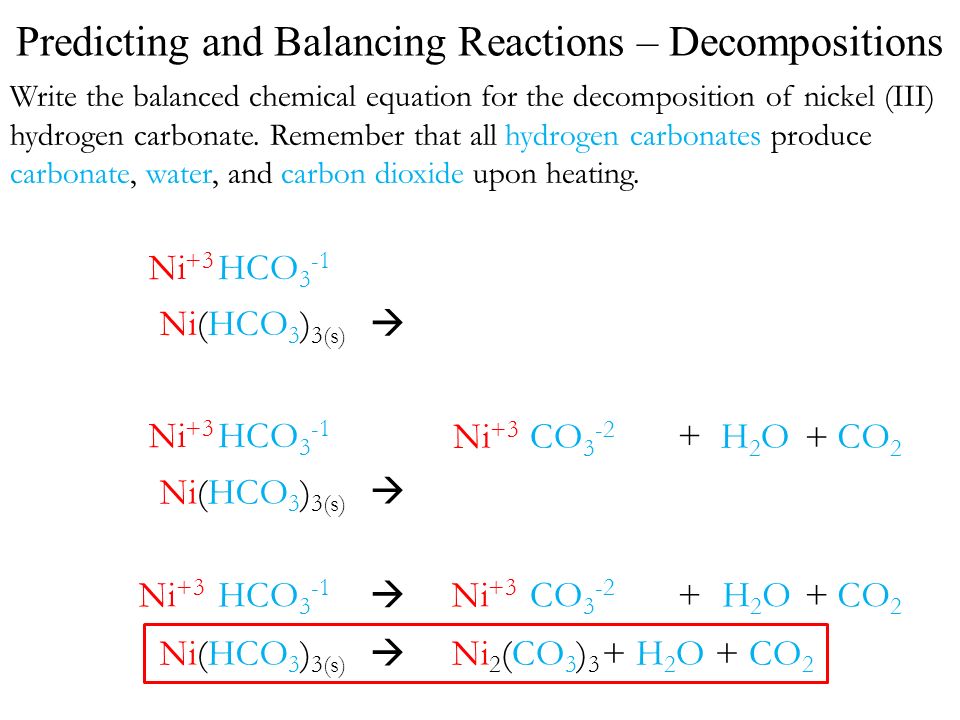
Double Replacement 1. Hydrogen sulfide is bubbled through a solution of silver nitrate. H 2 S(g) + AgNO 3 (aq) --> HNO 3 (aq) + Ag 2 S(s) H 2 S + Ag + - ppt download

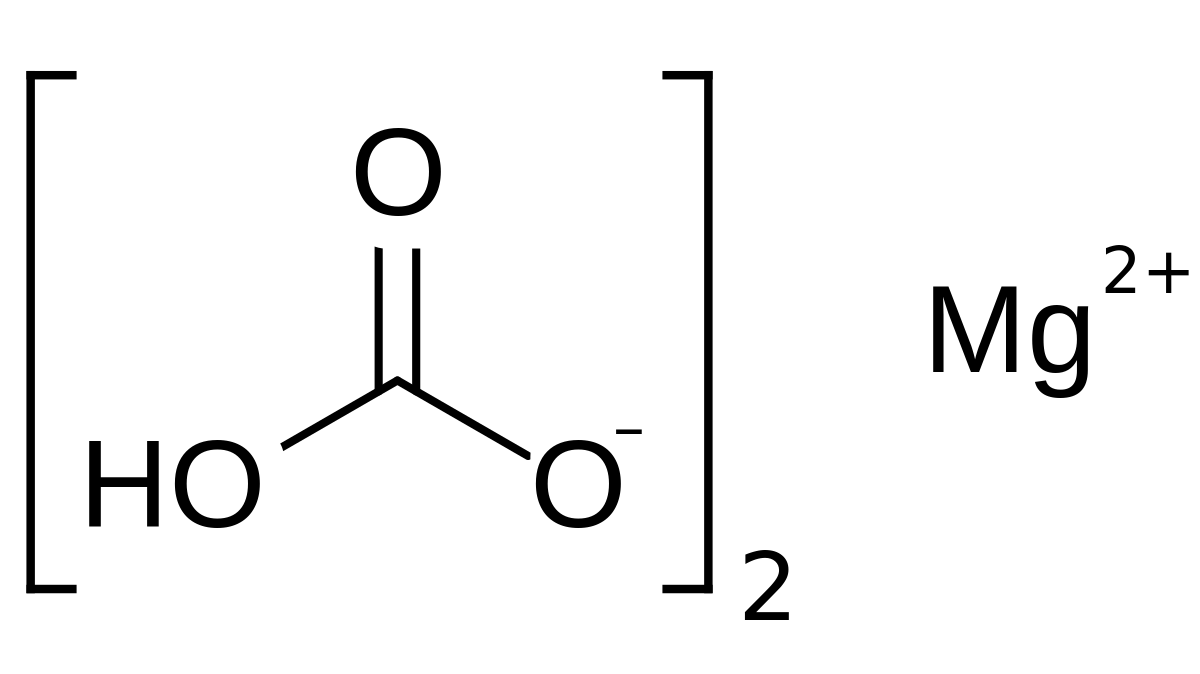
inorganic chemistry - Are lithium bicarbonate and magnesium bicarbonate both soluble in water? - Chemistry Stack Exchange
.jpg)
Sodium Bromide|Calcium Bromide|Ammonium Bromide|Lithium Bromide-Weifang Taihexing Chemical Industry Co.,Ltd.

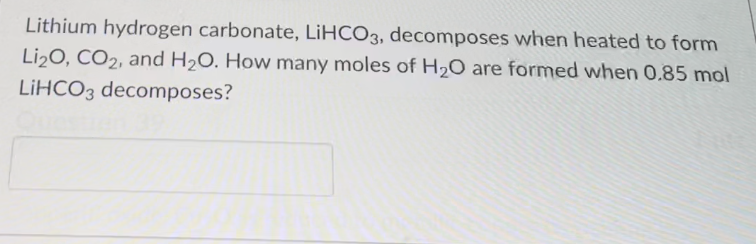
SOLVED: At the end of the reaction the solution contains the following particles: (Select all that apply:) carbonate ions carbonic acid molecules hydrogen carbonate ions hydronium ions hydroxide ions lithium hydrogen carbonate
Hierarchically structured lithium titanate for ultrafast charging in long-life high capacity batteries | Nature Communications

SOLVED: During used conbbusten [o fudi Vatlely Grlbg (0 pnrio Jiealing pierarEyy Eeckuifiliannt Ureaclion; carbon doxide end Kalter ae pprocuuced; (1O points BBellancet the reaction: CsHs @) - 01( C02 @ HhO
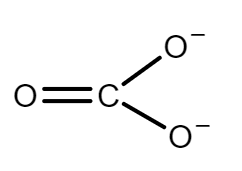
In what respects does lithium hydrogen carbonate differ from other alkali metal hydrogen carbonates?

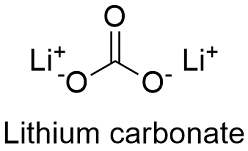
SOLVED: Decomposing Baking Soda EXPERIMENT DATE NAME SECTION POSTLABORATORY ASSIGNMENT 4 1.225- lithium carbonate hiumplarbopate !hiculahychrogen carbonate is decomposed 0.650 Calculate the theoretical yield and heating (0 produce percent yield of LizCO;.