Heterogeneous reaction of SO2 on CaCO3 particles: Different impacts of NO2 and acetic acid on the sulfite and sulfate formation - ScienceDirect

Steps involved in the synthesis of calcium carbonate nanoparticles from... | Download Scientific Diagram
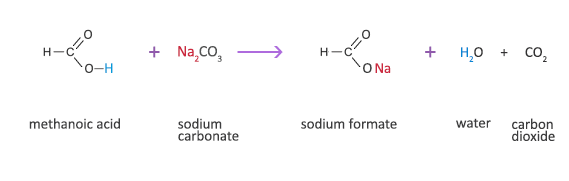
Explain why does the lime water turns milky in the reaction of acetic acid with sodium carbonate. - Sarthaks eConnect | Largest Online Education Community
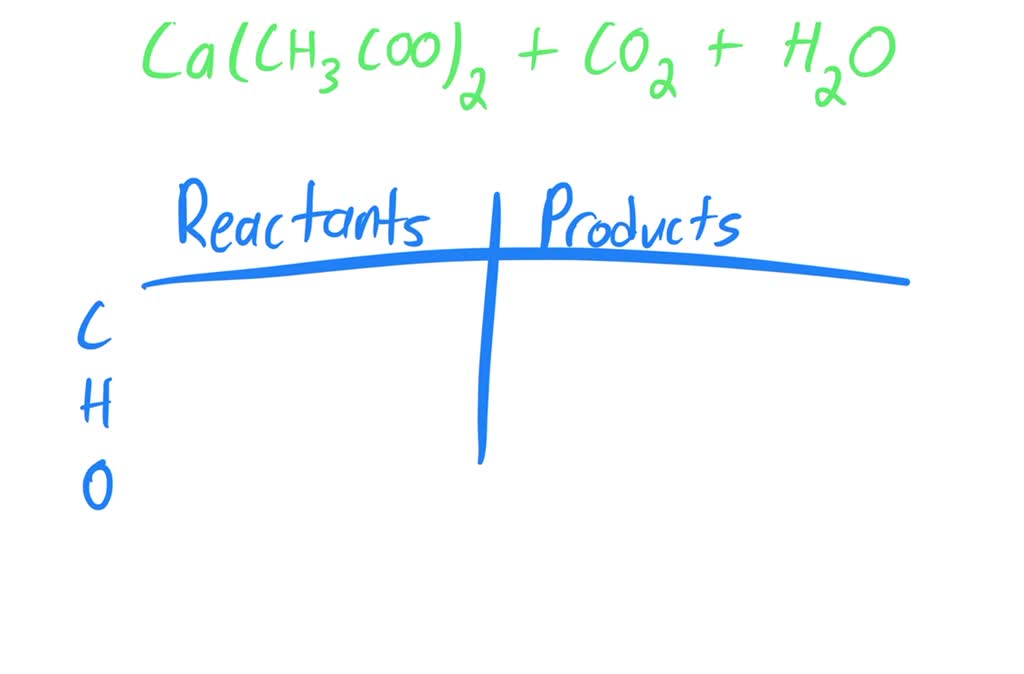
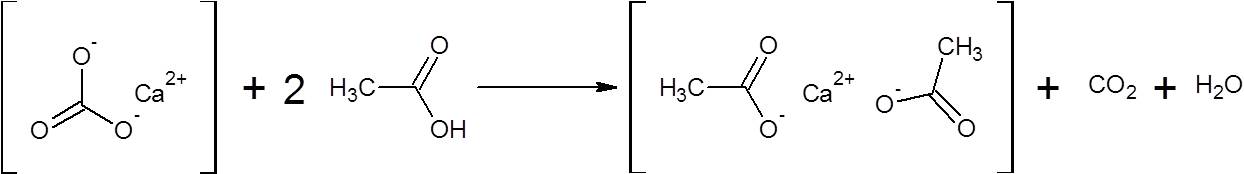
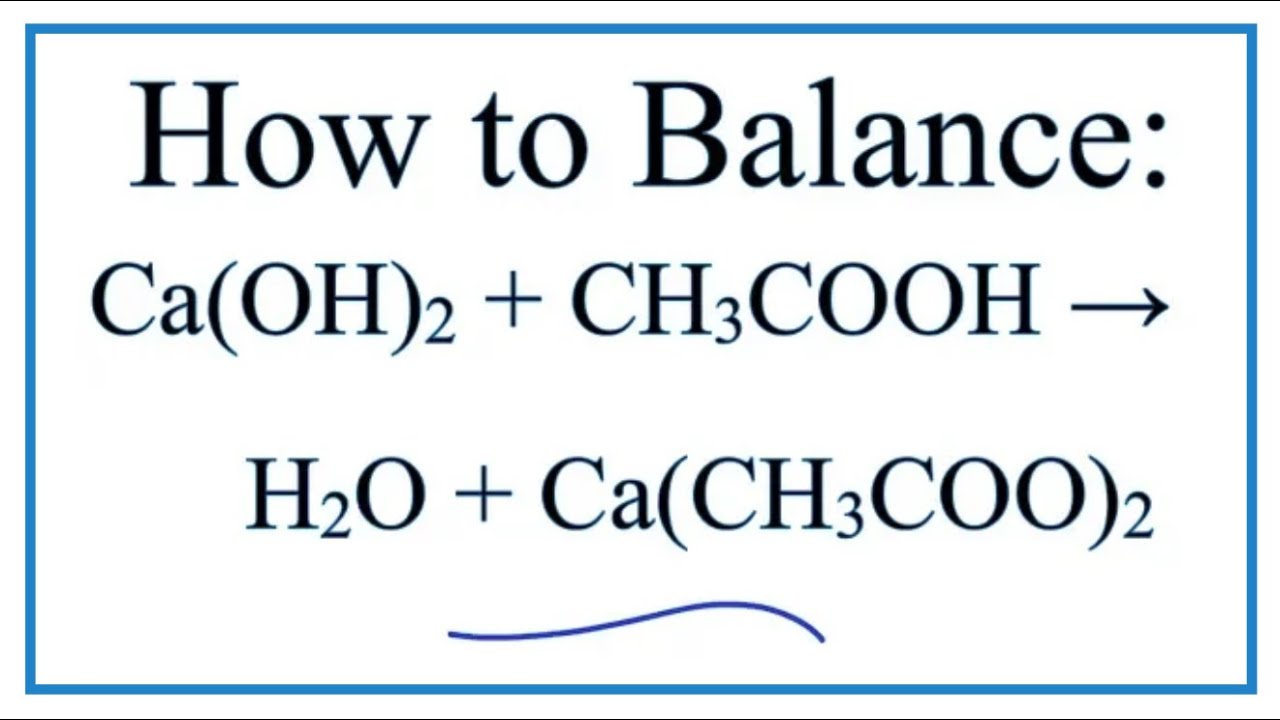
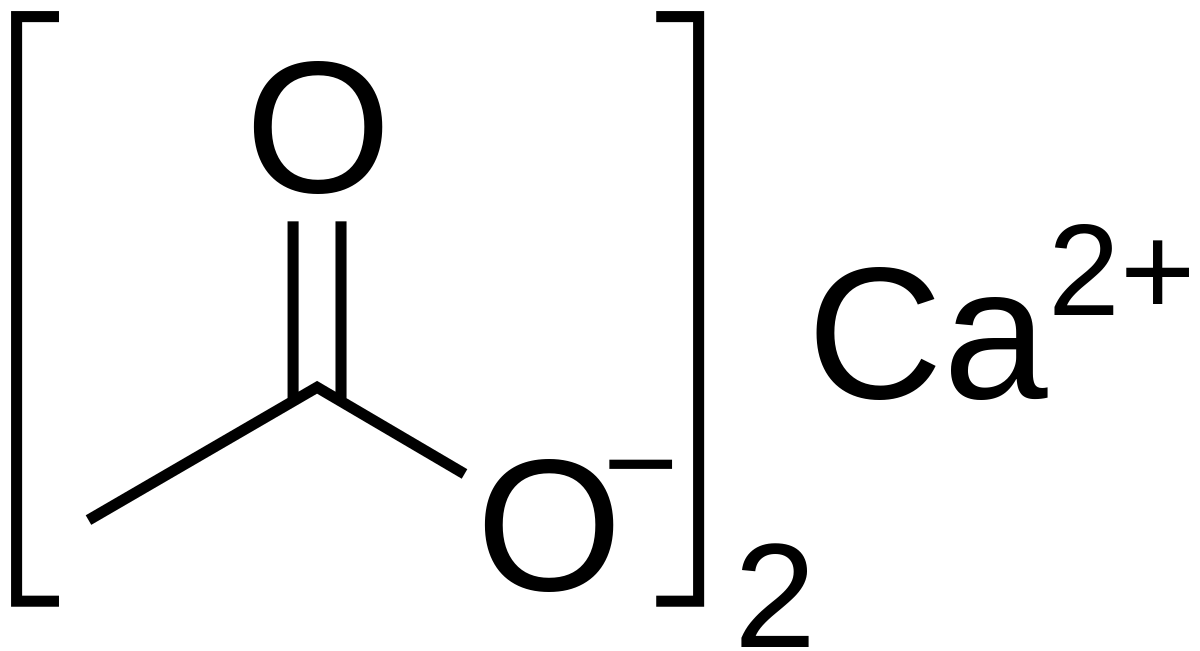
SOLVED: Find the limiting reagent in the reaction between calcium carbonate and acetic acid. This involves writing the balanced chemical equation between calcium carbonate and acetic acid, and finding the moles of

Question Video: Writing a Net Ionic Equation for the Reaction of Solid Calcium Carbonate with a Hydrochloric Acid Solution | Nagwa