SOLVED: Question 7 (1 point) Sodium carbonate reacts with nitric acid to form sodium nitrate; carbon dioxide, and water according to the balanced reaction below How many grams of sodium carbonate are

Question Video: Writing a Net Ionic Equation for the Reaction of Solid Calcium Carbonate with a Hydrochloric Acid Solution | Nagwa

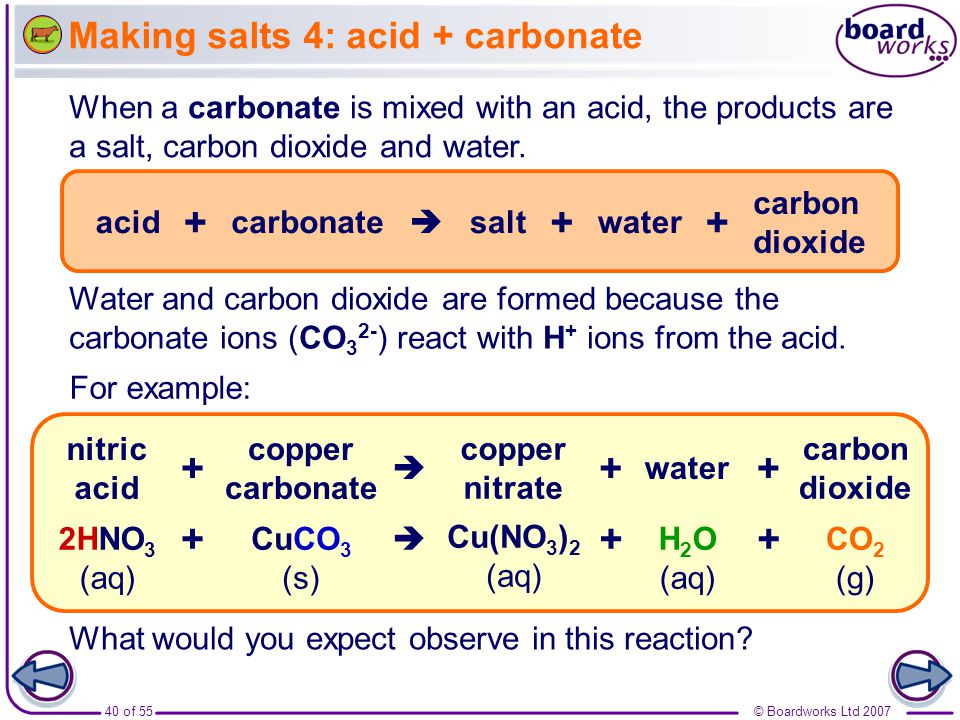
Write symbolic representation for the word equations and balance Sodium carbonate + Hydrochloric acid (dil) to Sodium chloride + Water + Carbon dioxide

pKa Ka data factors affecting Acidic reactions of carboxylic acids with metals oxides hydroxides carbonates hydrogencarbonate test advanced A level organic chemistry revision notes doc brown

Chemistry – Metal carbonate and hygrodencarbonates - Acids, bases and salts - Part 2 -English - YouTube

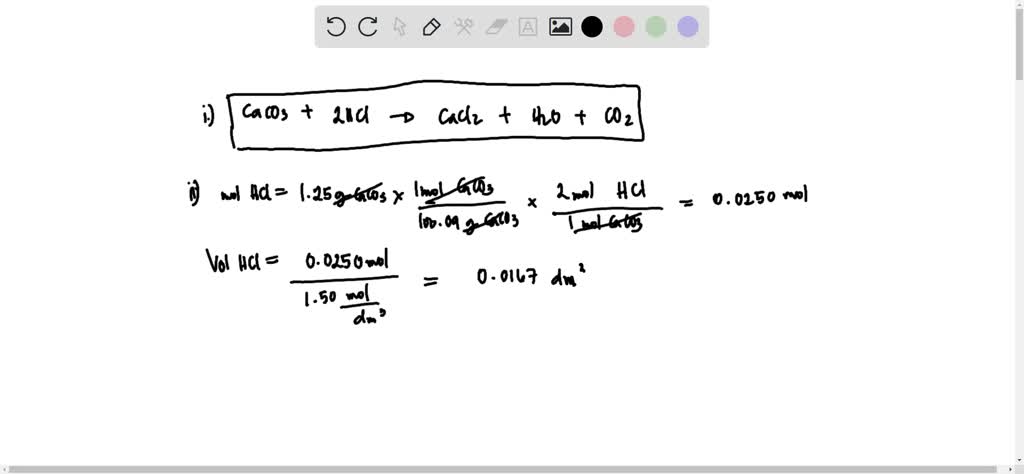
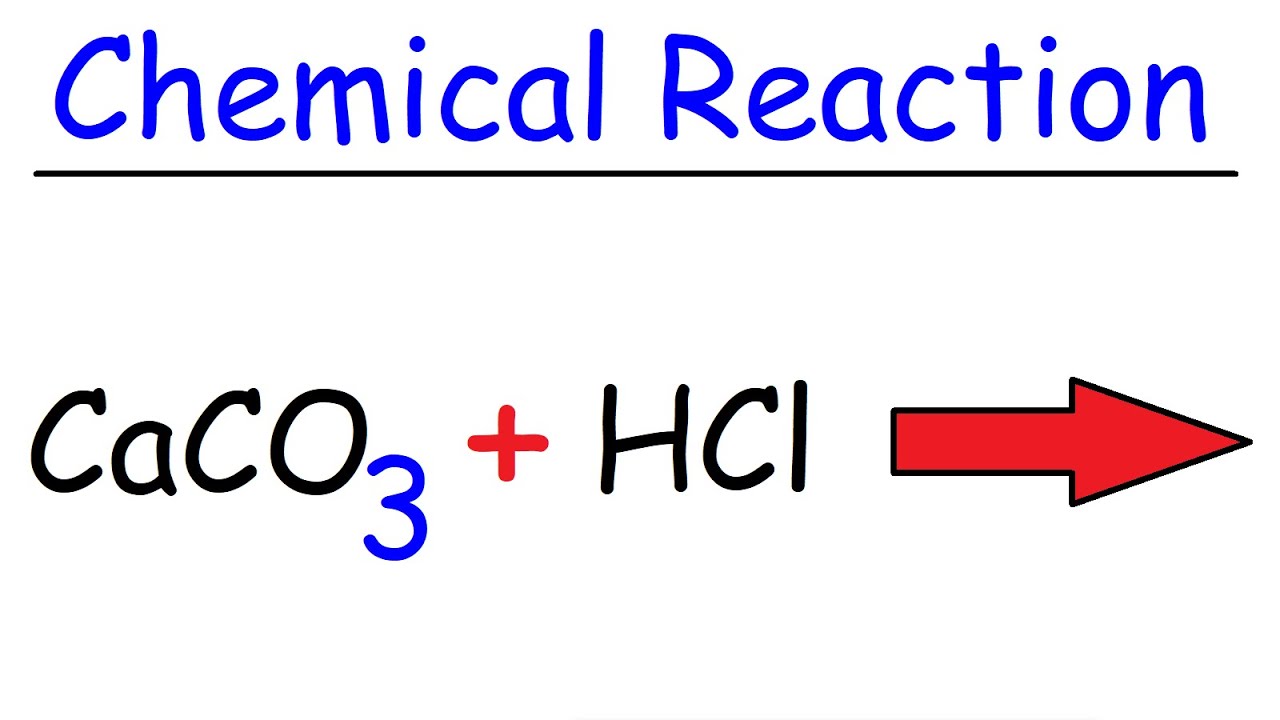
Calcium carbonate reacts with aqueous HCl to give CaCl2 and CO2 according to the reaction, CaCO3 (s) + 2HCl (aq) →CaCl2 (aq) + CO2 (g) + H2O (l) .What mass of CaCO3

Advanced Amino Acid-Based Technologies for CO2 Capture: A Review | Industrial & Engineering Chemistry Research

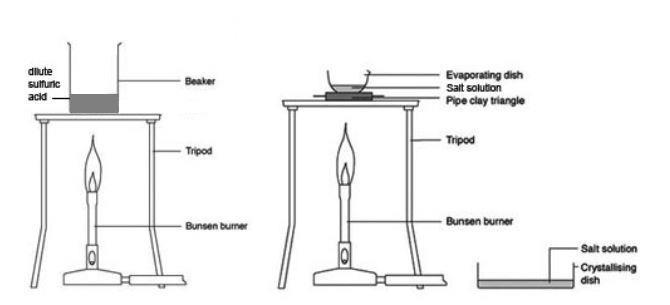
Acids and Metals Acids react with certain metals to produce hydrogen gas and the metal salt. metal acid metal salt. - ppt download