What precipitate will form when aqueous solutions of sodium carbonate calcium Na_2CO_3 and calcium chloride CaCl_2 are mixed? | Socratic

Calcium carbonate reacts with aqueous HCl to give CaCl2 and CO2 according to the reaction, CaCO3 (s) + 2HCl (aq) →CaCl2 (aq) + CO2 (g) + H2O (l) .What mass of CaCO3

Question Video: Writing a Net Ionic Equation for the Reaction of Solid Calcium Carbonate with a Hydrochloric Acid Solution | Nagwa

Calcium carbonate reacts with aqueous `HCl` to give `CaCl_(2)` and `CO_(2)` according to the rea... - YouTube

Figure 4. Variation of molar solubility for calcium carbonate (S) and pH as a function of the concentration of CO2(g) in ppmv. : Advanced Treatment of Chemical Equilibria in the Prediction of
Eco-friendly processes for the synthesis of amorphous calcium carbonate nanoparticles in ethanol and their stabilisation in aqueous media - Green Chemistry (RSC Publishing)

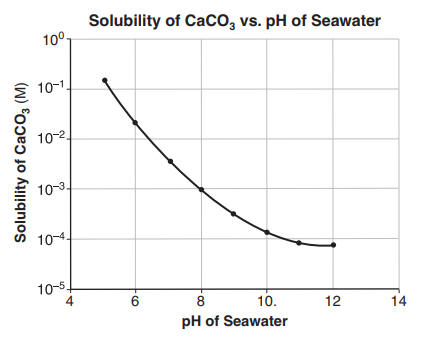
Solubility of calcium carbonate (lime scale) in water as a function of pH. | Download Scientific Diagram

The Effect of Monoethylene Glycol on Calcium Carbonate Solubility at High Temperatures | Industrial & Engineering Chemistry Research


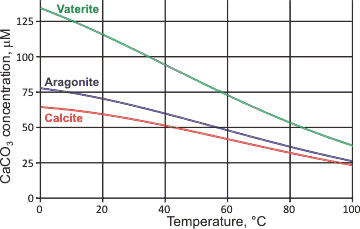
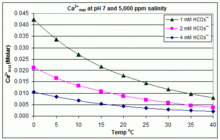
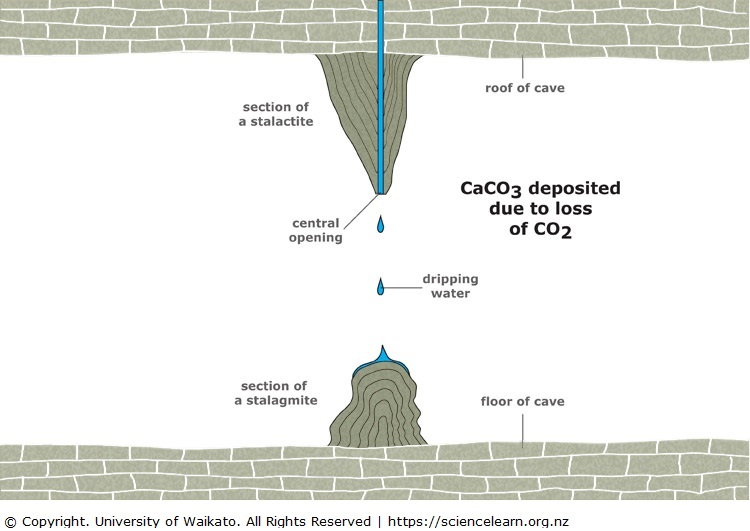
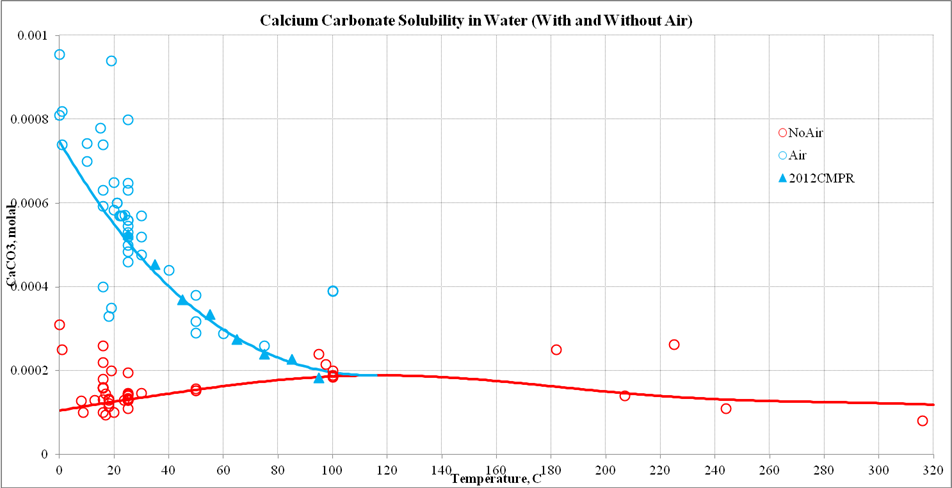
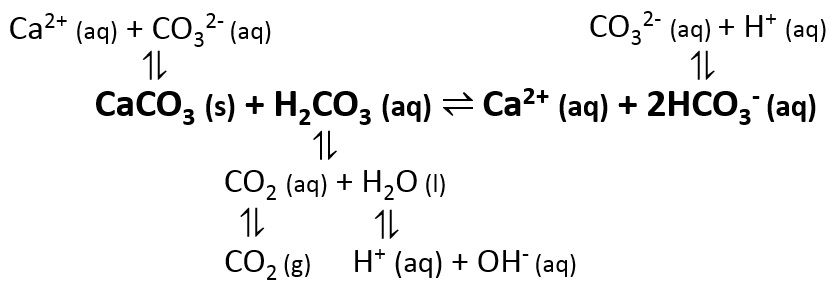
![Calcium carbonate solubility in function of pH [after (13)] | Download Scientific Diagram Calcium carbonate solubility in function of pH [after (13)] | Download Scientific Diagram](https://www.researchgate.net/publication/287707021/figure/fig8/AS:437295074549766@1481270678640/Calcium-carbonate-solubility-in-function-of-pH-after-13.png)