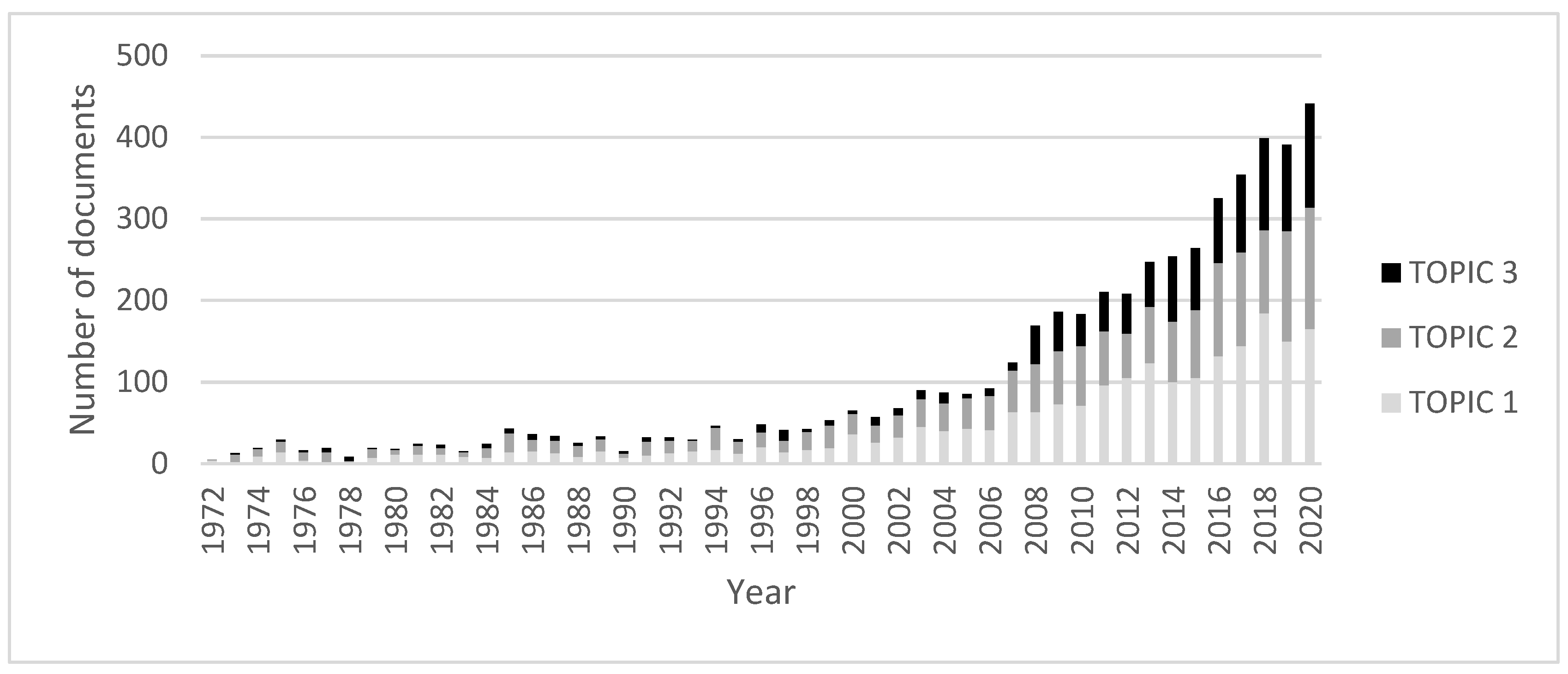
Water | Free Full-Text | Review of Techniques to Reduce and Prevent Carbonate Scale. Prospecting in Water Treatment by Magnetism and Electromagnetism
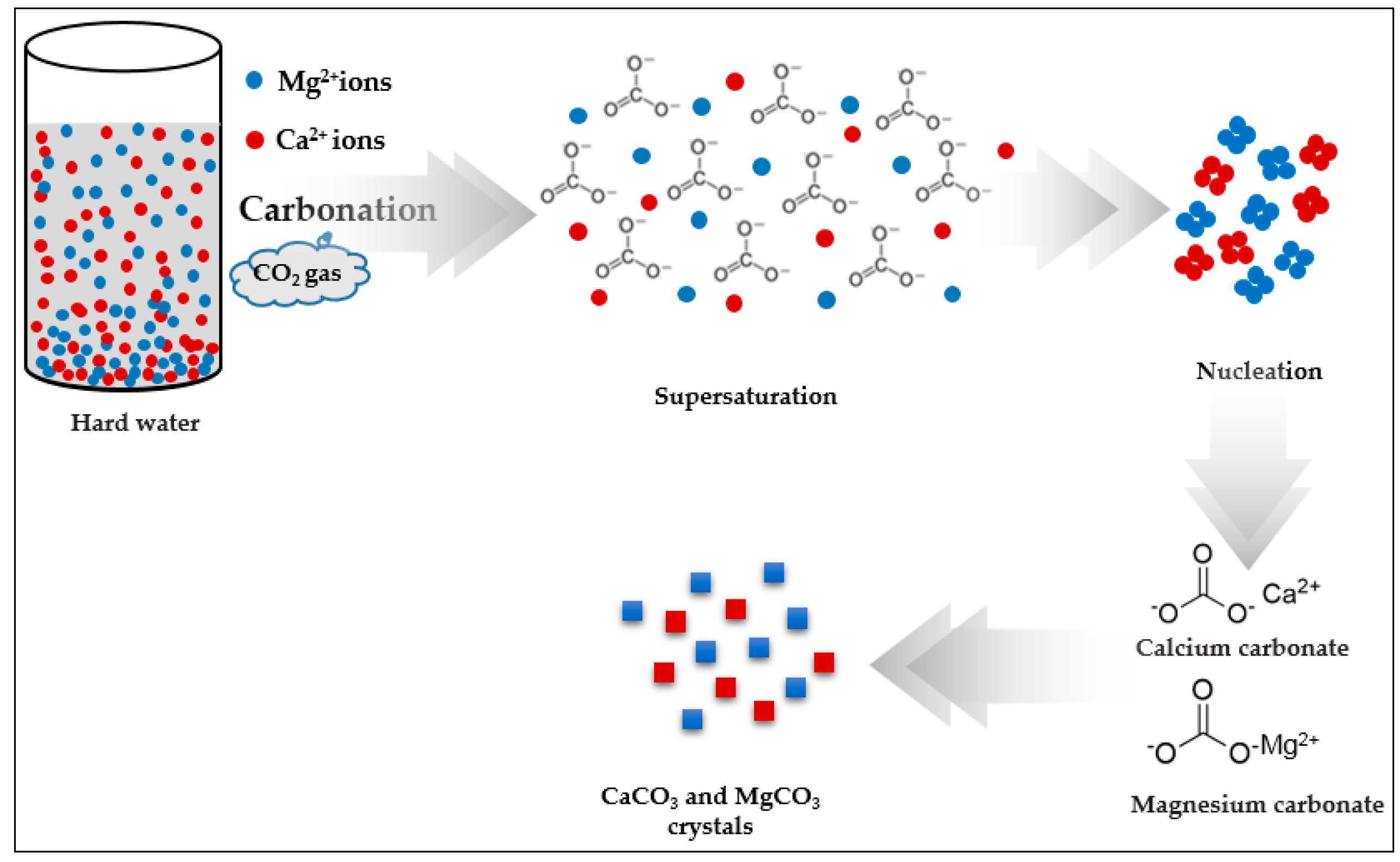
Water | Free Full-Text | Removal of Hardness from Water Samples by a Carbonation Process with a Closed Pressure Reactor

APEC PHPLUSKIT-14 US MADE 10" Alkaline High Purity pH+ Calcium Carbonate Inline Filter Kit with 1/4" Quick Connect : Amazon.in: Home Improvement

Assessment of calculation methods for calcium carbonate saturation in drinking water for DIN 38404-10 compliance – topic of research paper in Chemical sciences. Download scholarly article PDF and read for free on

The new Body Attack Natural Sports Water is a mineral water that quenches the thirst and contains 0.079 g calcium, 0.175 g hydrogen carbonate, 0.038 g chloride, 0.003 g of magnesium and

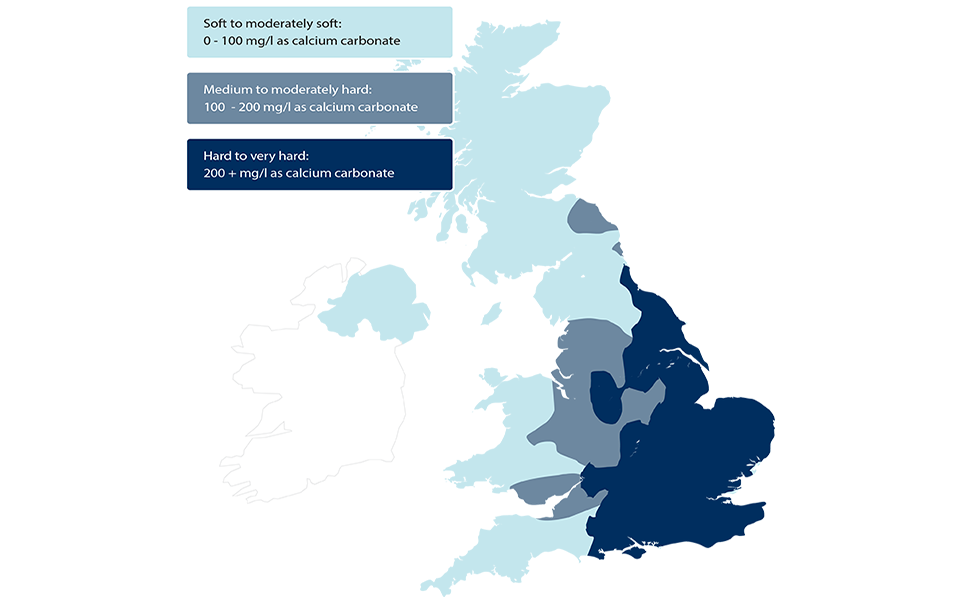
Milton Keynes has some of the hardest water in the UK - here's what it means for you | Milton Keynes Citizen

PDF) Assessment of calculation methods for calcium carbonate saturation in drinking water for DIN 38404-10 compliance