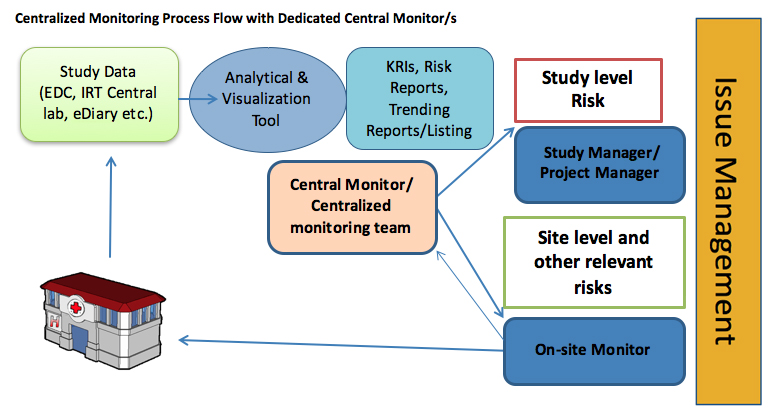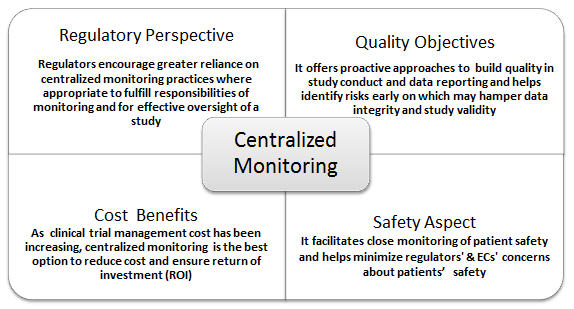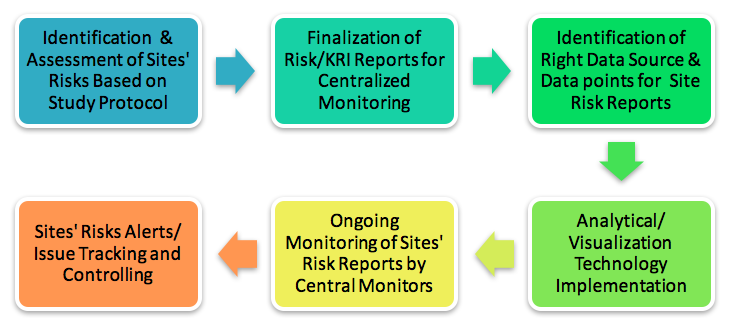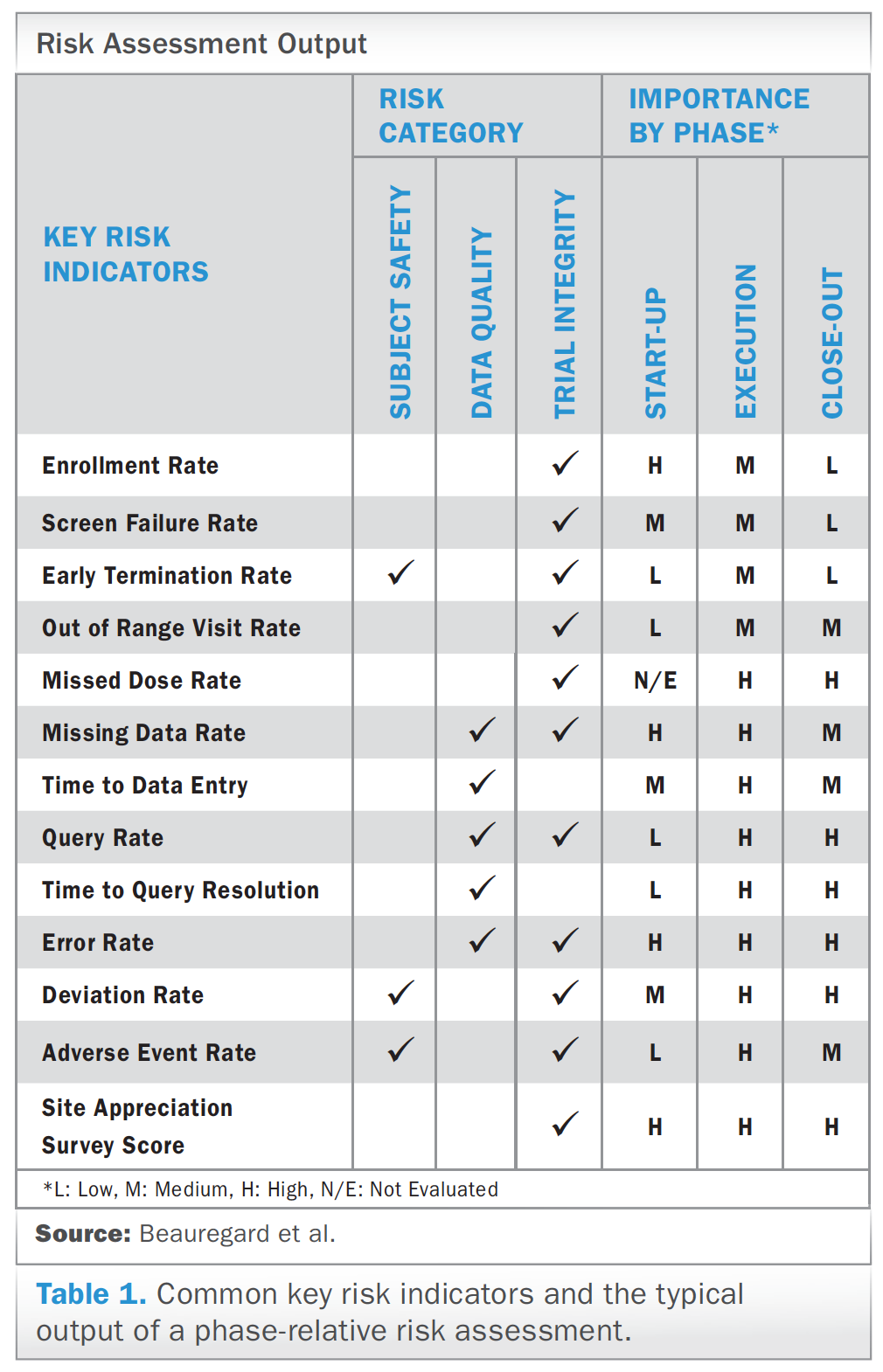
Central Monitoring Services(CenMon) | MakroCareRegulatory, Clinical Consulting Services to Biopharma & Medical Device Companies | MakroCare

ICT-based centralized clinical trial monitoring system. CTMS clinical... | Download Scientific Diagram
Centralized Monitoring in Practice — Lessons Learned from Early Implementers | 2019-01-21 | CenterWatch
Innovative approaches to investigator-initiated, multicentre paediatric clinical trials in Canada | BMJ Open

Integration of the Xcellerate risk and issue management system into the... | Download Scientific Diagram

Central Monitoring Is the Key to Data Integrity in Decentralized Clinical Trials - Medidata Solutions