A magnesium ribbon is burnt in oxygen to give white compound X accompanied by emission of light. - Sarthaks eConnect | Largest Online Education Community

What is a redox reaction? When a magnesium ribbon burns in air with a dazzling flame and forms - YouTube

5. Write word equations and then balanced equations for the reaction taking place when:(a) Dilute Su - YouTube
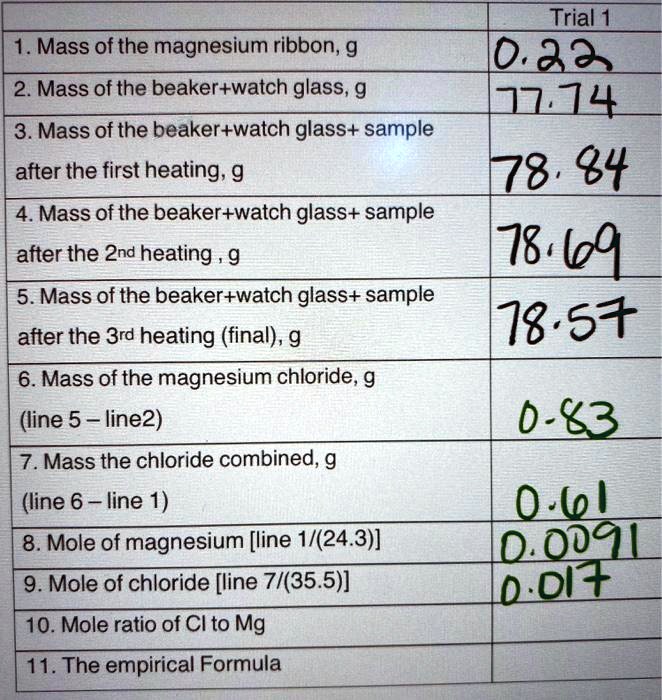
SOLVED: Trial 1 0.33 114 78.84 78 1g9 18.57 1. Mass of the magnesium ribbon, g 2 Mass of the beaker+watch glass, g 3.Mass of the beaker+watch glass+ sample after the first
Activity & Practical Of Burning Magnesium ribbon in a Bunsen flam | Class 10th & Class 9th level | edugrown – EduGrown School

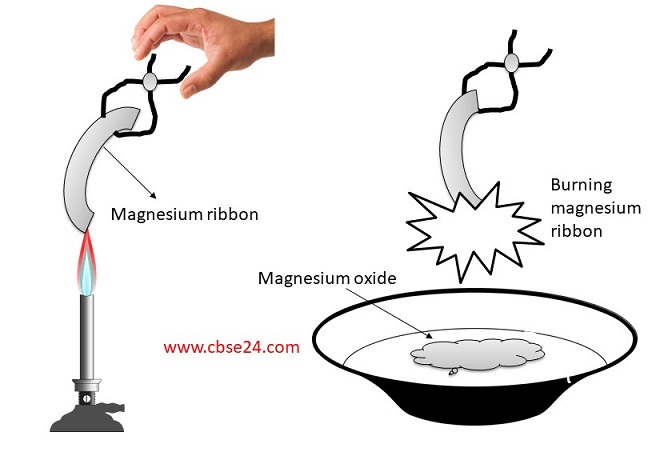
Magnesium ribbon burns with a dazzling flame in air (or oxygen) and changes to white substance magnesium oxide. Is magnesium being oxidised or reduced in this reaction ?

Show the dissociation of the following compounds on dissolving in water, with the help of chemical equation and write whether the proportion of dissociation is small or large.Hydrochloric acid, Sodium chloride, Potassium

A magnesium ribbon is burnt in oxygen to give a white compound 'X' accompanied by emission of light. If the burning ribbon is now placed in an atmosphere of nitrogen, it continues

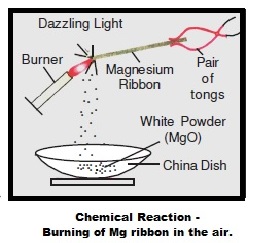
A student performs an experiment of burning magnesium ribbon in the air. A chemical reaction takes place and as a result, a white powder X forms along with a bright white light.The

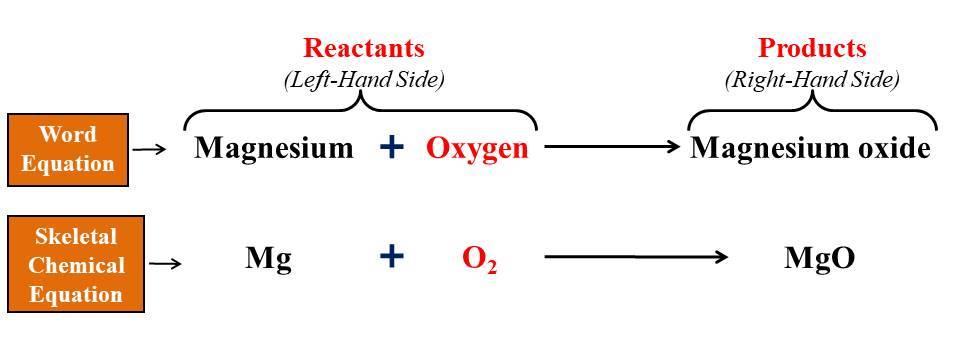
Chemical Equations. When chemicals are added together and a reaction occurs a chemical equation can be written The reactants go on the left of the equation. - ppt download
A 3.250g sample of magnesium is burned in a container of 12.500g oxygen. What mass of oxygen gas remains unreacted after the magnesium has been completely consumed to form magnesium oxide as





![Chemistry Class 10] Which is the correct observation of the reaction Chemistry Class 10] Which is the correct observation of the reaction](https://d1avenlh0i1xmr.cloudfront.net/410935ed-f4af-486a-8038-b5e1d6b7444e/burning-of-magnesium-ribbon---teachoo.jpg)