
Sodium Carbonate + Hydrochloric Acid - Na2CO3 + HCl - Molecular Equations & Net Ionic Equations - YouTube

Question Video: Determining the Products Formed from the Reaction between Sodium Carbonate and Hydrochloric Acid | Nagwa

SOLVED: Hydrochloric acid (HCl) reacts with sodium carbonate (Na2CO3), forming sodium chloride (NaCl), water (H2O), and carbon dioxide (CO2). This equation is balanced as written: 2HCl(aq)+Na2CO3(aq)→2NaCl(aq)+H2O(l)+CO2(g) Part A What volume of 1.75
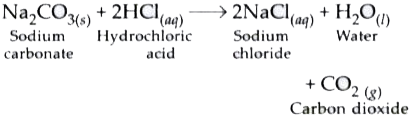
Write a balanced chemical equation for the reaction between sodium carbonate and hydrochloric acid indicating the physical state of the reactants and the products.
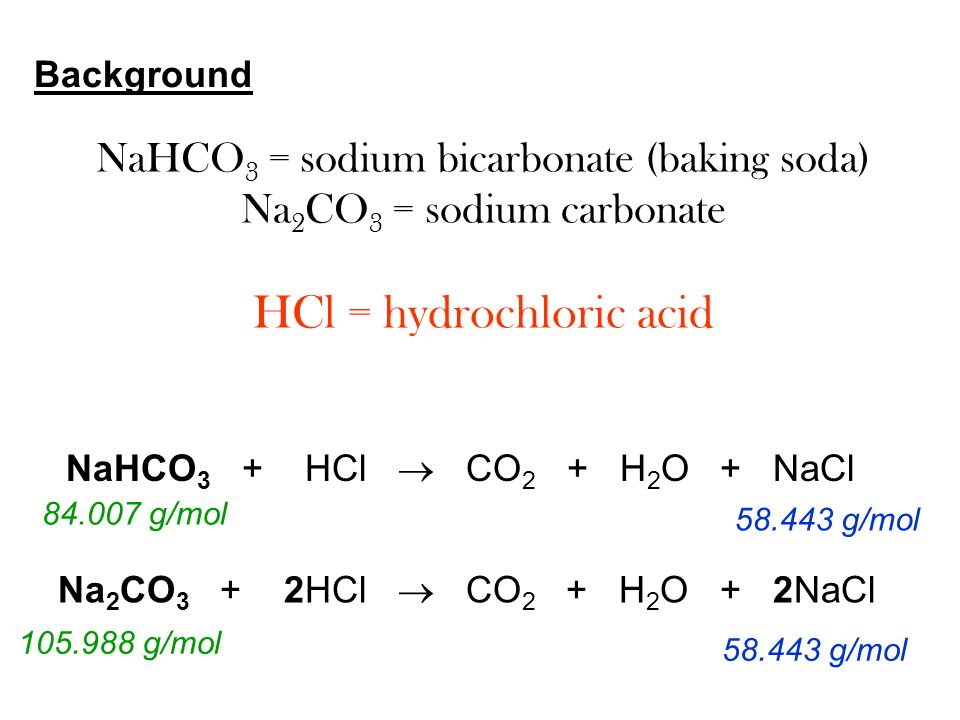
Lab 8 Sodium Carbonate or Sodium Bicarbonate? Objective To determine a compound to be either Na 2 CO 3 or NaHCO ppt download

Question Video: Writing a Net Ionic Equation for the Reaction of Solid Calcium Carbonate with a Hydrochloric Acid Solution | Nagwa

Molarity and Stoichiometry. 1.Calculate the number of grams of sodium carbonate that are required to react fully with mL of M HCl. Na 2 CO. - ppt download
Explain how a pH meter could be used to find the exact volume of acid required to completely react with a sodium carbonate solution? - Quora

a). What happens when dilute hydrochloric acid is added to sodium carbonate? Write a balanced - YouTube

SOLVED: 43. Samples of sodium carbonate powder were reacted with separate samples of excess hydrochloric acid: NazCOz (s) 2HCI (aq) COz (g) + ZNaCl (aq) + HzO (U) Reaction I: 1.0 g
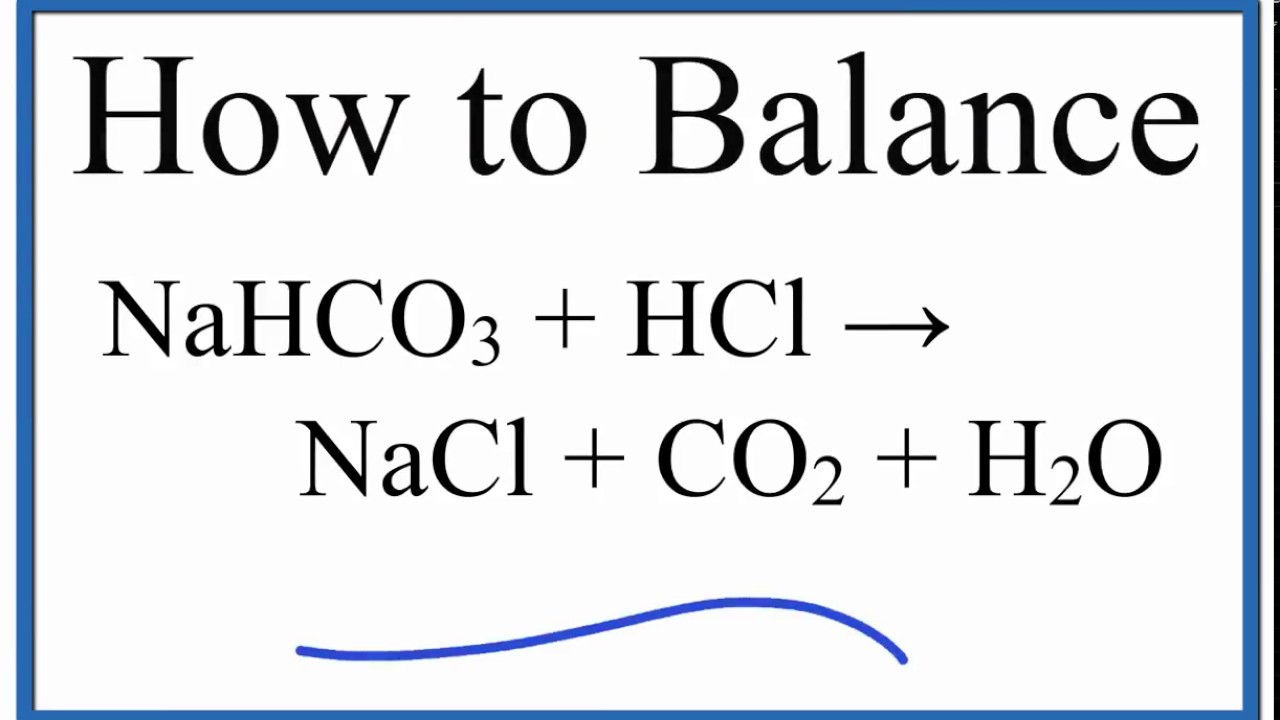
How to Balance NaHCO3 + HCl = NaCl + CO2 + H2O (sodium bicarbonate plus hydrochloric acid) - YouTube

During the titration of sodium carbonate with H Cl, the dissolved carbonate ion will exist in three different forms; CO_3^{-2}, H CO_3^{-1}, and H_2 CO_3. During which part of the titration (initial,

Sodium Carbonate + Hydrochloric Acid - Balanced Molecular and Net Ionic Equation - Na2CO3 + HCl | Quizalize








